Gore Ventures
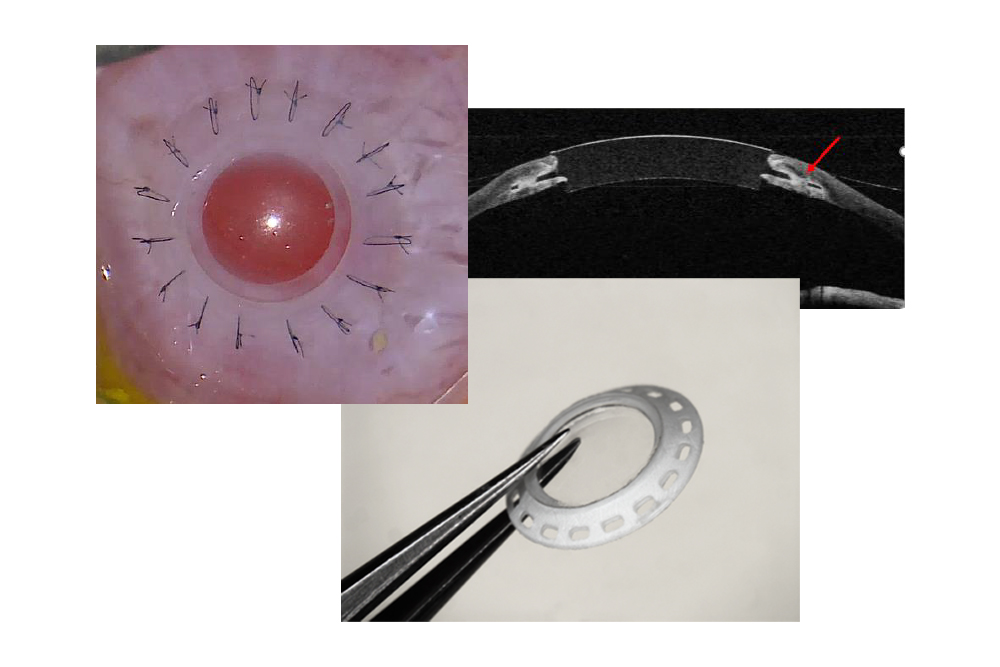
Investigational Synthetic Cornea Device
Exploring innovative surgical devices to treat global corneal blindness.
Developing an Investigational Synthetic Cornea Device as an Alternative to Human Donor Cornea Tissue, Aimed at Replacing an Opaque Cornea—the Fourth Leading Cause of Blindness Worldwide.
- A flexible and transparent fluoropolymer optic combined with a porous, biocompatible expanded PTFE (ePTFE) skirt that allows suturing of the device directly to the recipient eliminating the need for a carrier donor tissue
- Collaborated with leading keratoplasty surgeons and surgical centers to drive development1
- Currently being evaluated in a pilot clinical study2


Providing an Option When Donor Corneas Fail
Donor tissue transplantation is the standard treatment for corneal opacity, but it has limitations.
- Globally, 4–8 million people are blind due to corneal opacity. With only 1/70 individuals having access to donor corneas globally3
- Approximately 30% of donor corneas performed for all causes fail within the first 3 years of transplantation. This number increases considerably with each repeat transplantation4
- This investigational synthetic cornea device offers a potential solution:
- By eliminating the need for human donor corneas, thereby increasing the number of patients that can be treated.
- By increasing the chances of success due to lack of rejection or failure.
Thoughtfully Designed With Patient Care and Unmet Global Demand for Human Donor Corneas in Mind
Gore’s novel materials and innovative device design are intended to meet the critical need for a durable cornea device.
- Minimally invasive approach: Intended to require only a small central excision of the diseased cornea and does not alter the eye anatomy
- Suturable Design: Intended to eliminate reliance on carrier donor corneas for implantation
- Biointegration Potential: Intended for the recipient corneal tissue to adhere to the microporous material, minimizing the risk of retraction or infection
- Clear Optic: Expected to maintain transparency with built-in refractive power
Product Resources
- Surgical Video of Synthetic Cornea Device
- The Company Behind GORE-TEX® is Coming for your Eyeballs
- Design and Biocompatibility of a Novel, Flexible Artificial Cornea
- Prognostic Significance of Biointegration in Keratoprosthesis Implantation
- Twelve-Month Clinical and Histopathological Performance of a Novel Synthetic Cornea Device in Rabbit Model
References
- Dr. Anthony Aldave, UCLA; Or. Guillermo Amescua, Dr. Kathryn Colby, New York University; University of Miami; Or. Maria Cortina, University of Illinois, Chicago; Or. Jose de la Cruz, University of Illinois, Chicago; Dr. Jean Marie Parel, University of Miami
- Conde de la Valenciana, Mexico City, Mexico (Principal Investigators: Dr. Enrique Graue Hernandez, Dr. Arturo Ramirez Miranda)
- Gain P, Jullienne R, He Z, Aldossary M, Acquart S, Cognasse F, Thuret G. Global Survey of Corneal Transplantation and Eye Banking. JAMA Ophthalmol. 2016 Feb;134(2):167-73. doi: 10.1001/jamaophthalmol.2015.4776. PMID: 26633035.
- Ahmad S, Klawe J, Utine CA, Srikumaran D, Jimenez J, Akpek E. Survival of penetrating keratoplasty: a claims-based longitudinal analysis. Can J Ophthalmol. 2021 Feb;56(1):12-16. doi: 10.1016/j.jcjo.2020.07.019. Epub 2020 Sep 4. PMID: 32891566.
- Akpek EK, Aldave AJ, Amescua G, Colby KA, Cortina MS, de la Cruz J, Parel JA, Li G. Twelve-Month Clinical and Histopathological Performance of a Novel Synthetic Cornea Device in Rabbit Model. Transl Vis Sci Technol. 2023 Aug 1;12(8):9. doi: 10.1167/tvst.12.8.9. PMID: 37561510; PMCID: PMC10431210.Top of Form
Latest News
- / Events
Visit Us at World Cornea Congress IX
Visit Booth T-20 to explore our synthetic cornea device and connect with our team. Discover how we’re advancing vision restoration with cutting-edge innovation!
- / News
What is New in Artificial Corneas
Recently, reinvigorated efforts in developing fully synthetic flexible devices have resulted in several prototypes now undergoing clinical trials.
- / News
COFEPRIS Approves Clinical Trial for GORE Synthetic Cornea
The Federal Commission for Protection against Sanitary Risks (COFEPRIS) has recently authorized a pioneering clinical trial, which aims to assess the effectiveness of the GORE synthetic cornea device in patients suffering from corneal clarity loss.
- / News
What is New in Artificial Corneas
Since the first successful full thickness corneal transplantation was performed by Eduard Zirm in 1905, the procedure has grown to become the most commonly performed transplantation globally.
- / News
COFEPRIS Approves Clinical Trial for GORE Synthetic Cornea
The Federal Commission for Protection against Sanitary Risks (COFEPRIS) has recently authorized a pioneering clinical trial, which aims to assess the effectiveness of the GORE synthetic cornea device in patients suffering from corneal clarity loss.